Flowtron ACS900
Smart, intuitive and quiet with Compliance Monitoring
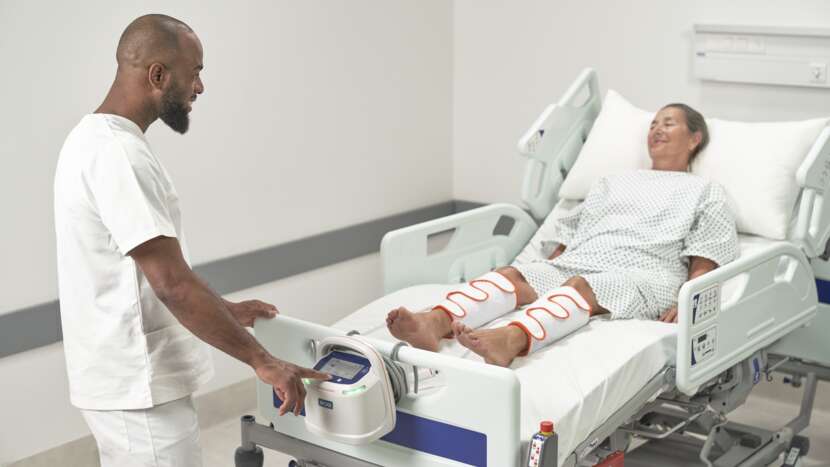
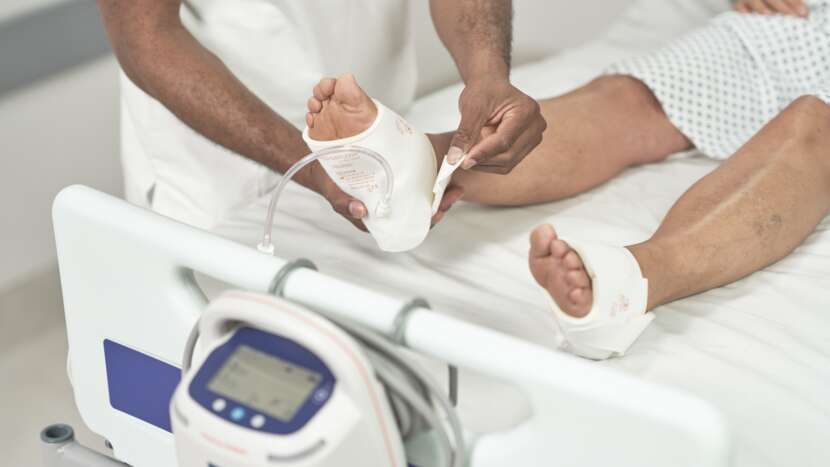
Reduce the risk of VTE in your facility with the Flowtron ACS900 pump, featuring SmartSense™ 2 Automatic Garment Recognition and Compliance Monitoring technology.
PROVEN EASE OF USE AND SAFETYDesigned to give you freedom of choice, the Flowtron system offers both uniform and sequential modes in one easy-to-use pump.
Flowtron Active Compression System is the safe, convenient and flexible way to deliver VTE prevention therapy.¹⁻⁴ At Arjo, we have built on the Flowtron legacy for decades, continuously improving our offering to ensure the best possible performance of IPC therapy across healthcare environments. We continuously strive to make everyday tasks easier for caregivers, and enable them to spend more time caring for their patients.
An evaluation conducted by the ECRI Institute, an independent non-profit organisation focused on identifying the safest and most effective solutions for care, rated the Flowtron ACS900 against a comparable Sequential Compression System in terms of performance, safety, workflow, patient experience and cost of ownership. For information on how to obtain a copy of the report, please contact your local Arjo representative.
SMARTSENSE 2 AUTOMATIC GARMENT RECOGNITION
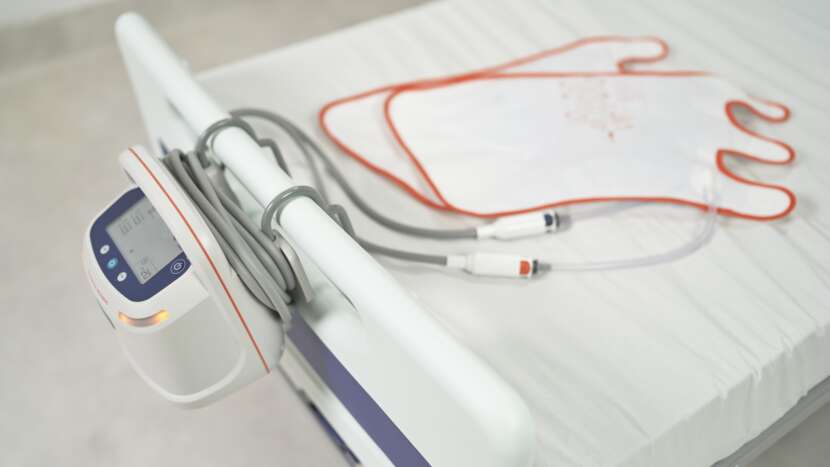
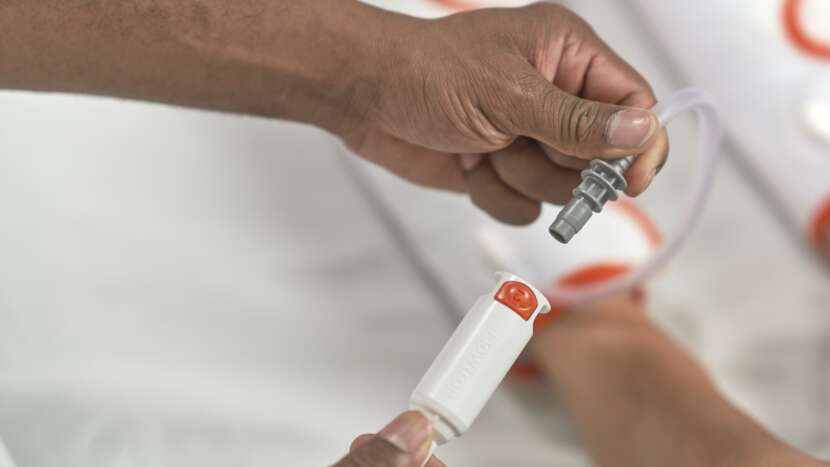
Arjo’s patented garment detection technology automatically sets the correct pressure and compression cycle, without the need for any additional user intervention. Simply attach the snap-lock connectors to the Flowtron ACS900 pump and the system easily and safely does the rest.
· Reducing the need for user intervention by automatically identifying garments connected and setting the correct pressure and compression cycle
· Designed to reduce the risk of patient discomfort and medical device-related pressure injury with a new low-profile connector design
· Reducing medical waste with a new, more environmentally sustainable, connector design using less plastic material
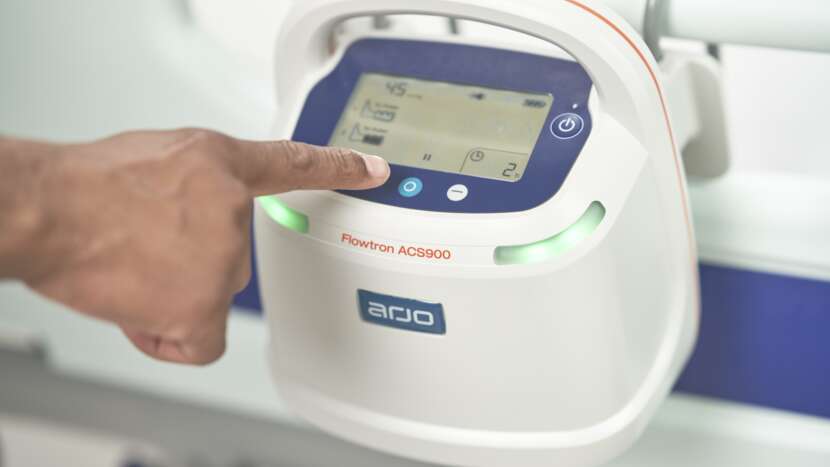
SMARTSENSE 2 COMPLIANCE MONITORING
To aid concordance and facilitate tracking and documentation of therapy, ACS900 provides intuitive compliance monitoring that detects garment wear-time during IPC. It allows the pump to record and display actual therapy as well as non-compliant time, and alert upon lack of adherence. This makes it easier for caregivers to ensure compliance with prescribed therapy and to enter accurate data into patient records.
· Supporting clinical outcome by helping to ensure effective delivery of IPC therapy
· Assisting caregiver intervention by alerting upon noncompliance to IPC therapy
· Aiding documentation of delivered therapy by providing data for patient records
REFERENCES:
1. Arjo Data on File: Summative Usability Validation Report 100035519. December 2014.
2. Arjo Data on File: Customer Acceptance Test (CAT) Report 100035688. June 2015.
3. Arjo Data on File: Functional Test Report 100035587. December 2019.
4. Arjo Data on File: Software Test Report 100035545. December 2019.
Automatic garment recognition together with one-button start make Flowtron a true plug-and-play solution that is easy to set up and operate. Reducing the need for user intervention
by automatically identifying garments connected and setting the correct compression profile – for safety and efficacy.1-3
COMPLIANCE MONITORING
Intuitive on-screen compliance monitoring, recording and displaying actual therapy as well as non-compliant time in an accurate manner. Alerts the caregiver upon garment removal to aid concordance, and facilitates tracking and documentation of IPC therapy.4-5
ALARMS AND INDICATORS
Advanced alarms, including visual indicators allowing operation to be clearly seen from any direction, and real-time pressure indication designed to limit the risk of operator error and potential patient harm – for safety and caregiver peace of mind.3
QUIET OPERATIONS
QuietConcept™ noise reduction technology significantly reducing pump noise to minimise patient and caregiver disruption. Allowing the ACS900 to deliver therapy in a quiet manner across all garment types, contributing to a quieter care environment.6
COMPLETE RANGE OF GARMENTS
A wide variety of garment types and sizes ensuring effective and comfortable therapy for all patients and clinical needs.3,7,8 The system allows for any combination of foot, calf and calf-and-thigh garments to be used simultaneously.
DURABLE AND ENERGY EFFICIENT
VTE prevention around the clock* with SmartEnergy™ enhanced power management ensuring uninterrupted calf therapy for a minimum of 24 hours when the pump is not connected to a power outlet9. SmartEnergy also contributes with reduced power consumption, CO2 emissions and cost.10
FIXED TUBESETS
Ready for therapy at all times with fixed tubesets preventing disconnection and loss of tubing, hence eliminating the inconvenience and cost of replacements.3
INTEGRATED CABLE MANAGEMENT SYSTEM
Integrated cable management system aiding tubeset and power cord management in the clinical area and during storage/transportation, promoting caregiver convenience and patient safety.1-3,11
REFERENCES:
1. Arjo Data on File: Customer Acceptance Test (CAT) Report 100035688. June 2015.
2. Arjo Data on File: Functional Test Report 100035587. December 2019.
3. Busby J, Holst K, Hansson K. User feedback on the Flowtron® ACS900 pump and Tri Pulse garment range. Arjo Whitepaper. June 2021. Arjo.A00491.1.0.INT.EN.
4. Arjo Data on File: Compliance Monitoring Test Report 100126883. November 2021.
5. Arjo Data on File: Summative Evaluation Report (SER) 100116863. November 2021.
6. Arjo Data on File: Audible Test Report 100127045. ACS900 MLU. Acoustic Noise Testing of Medical Pumps. Southwest Research Institute. November 2021.
7. Ellis J. The textile properties of Deep Vein Thrombosis (DVT) garments: a factor in patient compliance with Intermittent Pneumatic Compression (IPC) systems. Arjo Whitepaper. March 2019. Arjo.A00096.1.0.INT.EN.
8. Arjo Independent Test Data on File. Tri Pulse: water vapour resistance, thermal resistance (single plate method), drying time, liquid wicking rate and water vapour permeability testing. September 2019. Test report E-008677/C.
9. Arjo Data on File: Battery Study Report 100127047. ACS900 MLU. November 2021.
10. Arjo Data on File: Electrical Efficiency Measurements Report 100127046. ACS900 MLU.
11. Arjo Data on File: Summative Usability Validation Report 100035519. December 2014.
| Model | |
|---|---|
| ACS900 | Standard |
| ACS900 | OR (Operating Room) |
| Specifications | |
| Type of compression | Uniform or Sequential Calf, Calf/Thigh, Foot |
| Uniform: | 40 ± 5 mmHg |
| Sequential: | 45 ± 5 mmHg |
| Foot: | 130 ± 10 mmHg |
| Compression cycle Uniform/Sequential: | 12 sec. inflation; 48 sec. deflation |
| Foot: | 3 sec. inflation; 27 sec. deflation |
| ACS900 standard tube length | 2.1m/7ft |
| ACS900 OR tube length | 4.0m/13ft |
| Battery type | 14.4V, 2600mAh. Lithium Ion pack |
| Power | 100V-230V, 50-60Hz, 10-40VA |
| Pump size | H230 x W226 x D196mm / 9.1 x 8.9 x 7.7in |
| Weight | 3.8kg/8.5lb |
Explore this product
Type: Brochure
Type: Brochure
Type: Brochure
Type: Product specifications sheet
Type: Instructions for use (IFU)
Type: Instructions for use (IFU)
Type: Quick Reference Guide (QRG)
Type: Quick Reference Guide (QRG)
Type: Quick Reference Guide (QRG)
Type: Clinical summary and/or focus document
Type: Clinical summary and/or focus document
Type: Clinical summary and/or focus document
Type: Whitepaper
Type: Whitepaper
Type: Whitepaper
Type: Brochure
Type: Brochure
Type: Brochure
Type: Product specifications sheet
Type: Instructions for use (IFU)
Type: Instructions for use (IFU)
Type: Quick Reference Guide (QRG)
Type: Quick Reference Guide (QRG)
Type: Quick Reference Guide (QRG)
Type: Clinical summary and/or focus document
Type: Clinical summary and/or focus document
Type: Clinical summary and/or focus document
Type: Whitepaper
Type: Whitepaper
Type: Whitepaper