
Evenda – Design that Connects
Evenda is a long term care bed designed with mobility and cognitive care challenges in mind – to help residents and carers connect, and to help residents feel at home and connected to their environment.
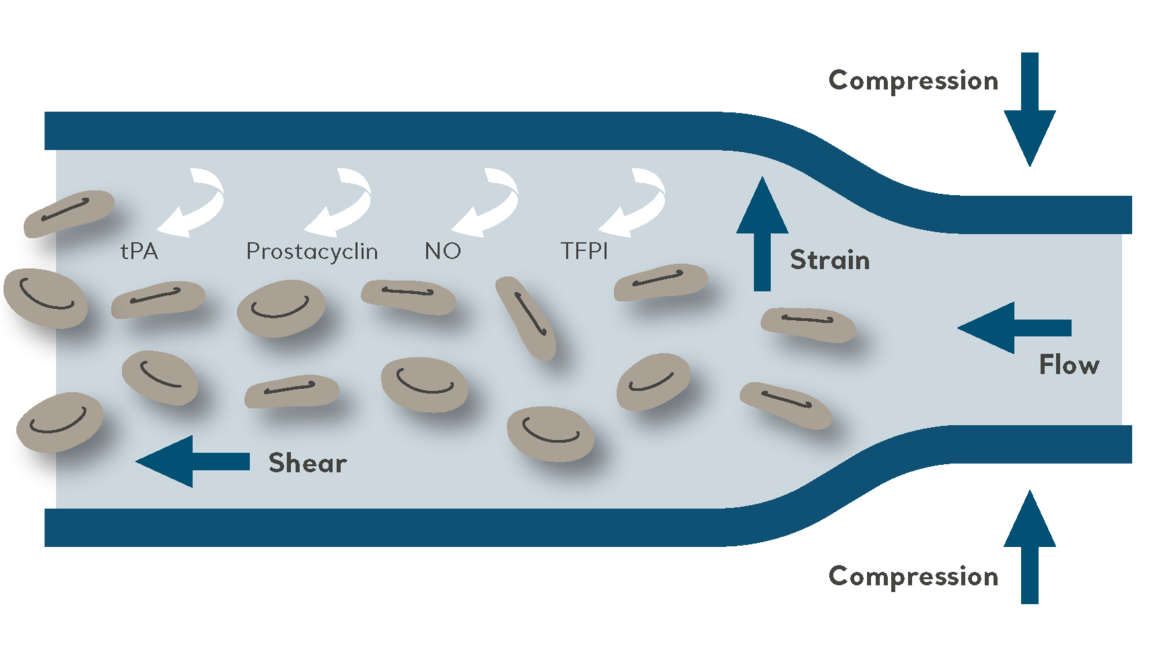
Understanding the Importance of Compression Therapy in VTE Prevention
For patients at risk of venous thromboembolism (VTE), intermittent pneumatic compression (IPC) is a clinically proven and well-established prevention method. Read our blog post to learn how it works.

''Movement provides a better quality of life for our residents''
For Benevit’s nursing homes, long-term sick leave time has dropped massively, partly due to ergonomics training and use of aids. One key differentiator: the integration of the Arjo MOVE program, which has shown positive impacts on physical and mental stress within the nursing teams, and a better quality of life for residents.
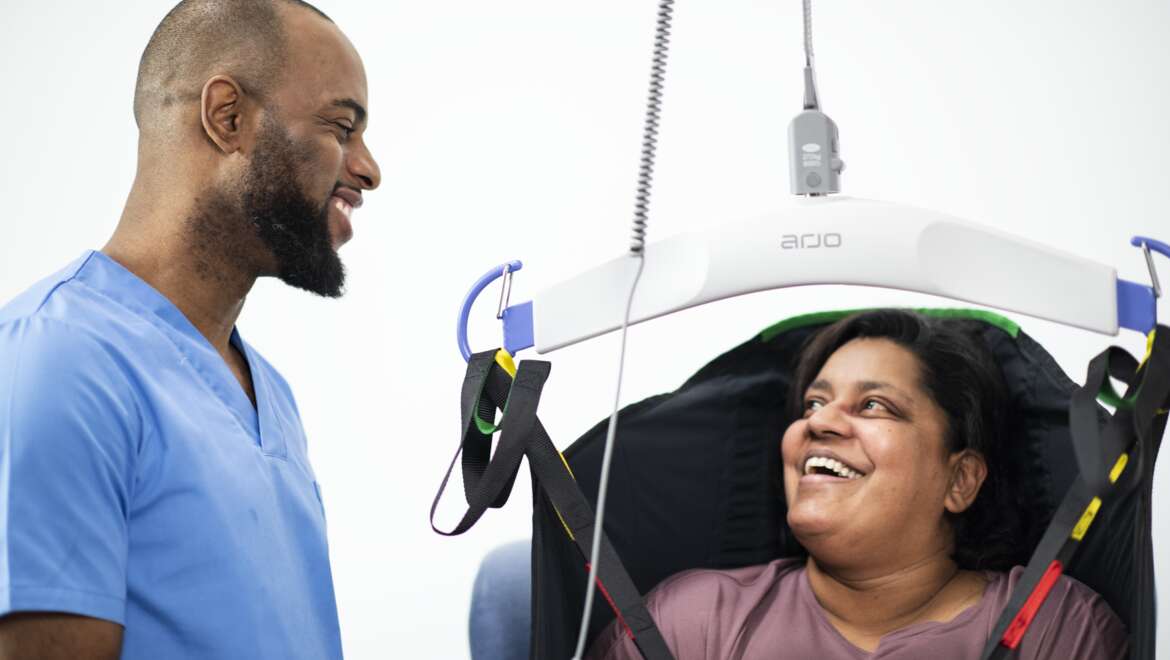
Do you consider these 7 factors in the patient mobility assessment process?
The International Standards for manual handling of people in the healthcare sector discuss various methods of assessment1. The most essential transfers take place multiple times throughout the daily journey.

Annual and Sustainability Report 2023
View our latest Annual and Sustainability Report

Introducing My Arjo: Your portal to Arjo tools and services
MyArjo is a personalised web portal providing access to Arjo tools and services with one secure login.
Sign up to get MyArjo access and begin your journey to Empower Movement today.
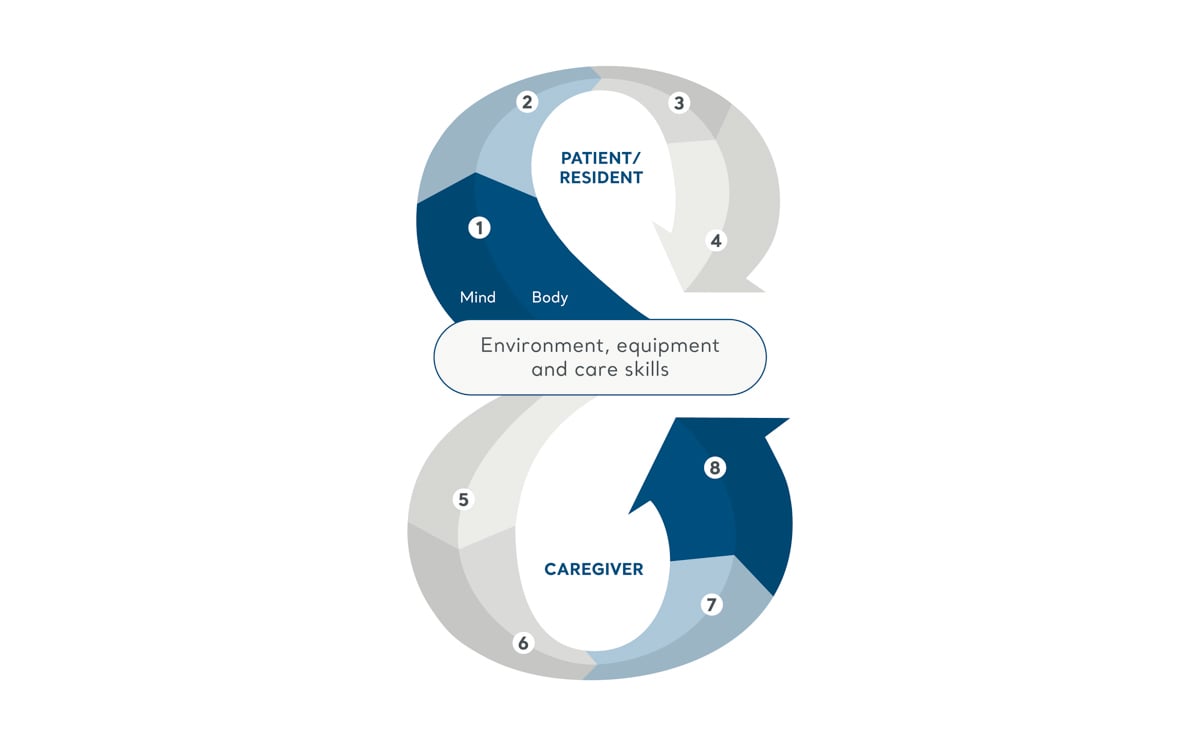
The Positive Eight Philosophy
Our care philosophy explains how facilitating mobility can catalyse a chain of benefits impacting quality of life, caregiver satisfaction, operational efficiency, and financial outcomes.

Mobility gallery
Based on 5 levels of mobility from A to E, the Arjo Mobility Gallery provides a basis for understanding how an individual can contribute to their mobility, as well as how to safeguard caregivers from injuries.

Arjo MOVE
Rooted in clinical evidence and driven by your facility data, Arjo MOVE programs arm you with the tools and knowledge to reach your clinical and operational goals.
Arjo at a glance
Facts about Arjo
6,500
employees globally
1957
Arjo was founded
100+ countries
where Arjo sells products and services
Empowering Movement
is at the heart of everything we do
We ensure value by
Preventing the impact of immobility
Promoting greater mobility with dignity
Protecting the wellbeing of caregivers
Optimising costs and efficiencies in care
Global news
Resolutions at Arjo’s Annual General Meeting 2024
Arjo’s interim report January-March 2024
Arjo announces date for 2024 Q1 report and conference call
Arjo publishes its Annual and Sustainability Report for 2023
NOTICE OF ANNUAL GENERAL MEETING IN ARJO AB (PUBL)



